
Illustration by Allan Lasser
How gut bacteria manipulates your immune system – by mimicking it
Scientists are discovering how microbes 'speak' with the body
Complex systems relay information through signals: markets have prices, computers have binary, humans have spoken and written language. The system works when the encoded and decoded signals reliably represent the agreed-upon meaning.
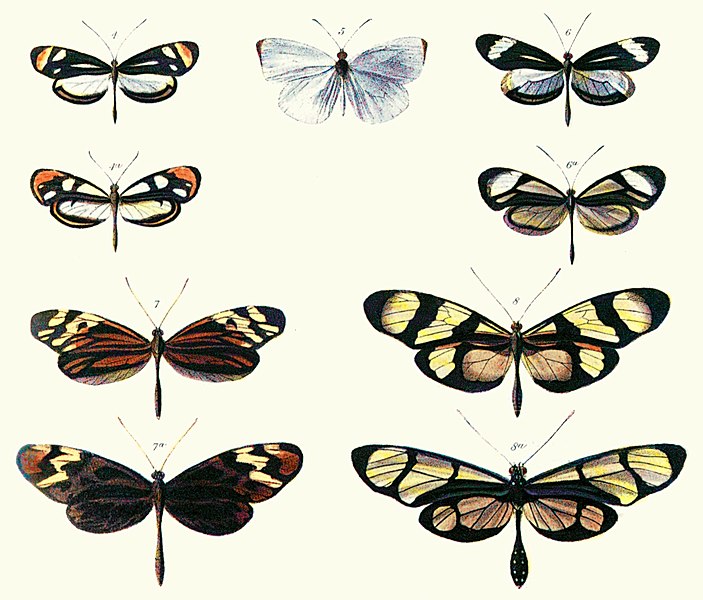
Within these systems, cheaters can thrive by sending dishonest signals: the harmless milk snake copies the coloration and patterned scales of the poisonous false coral snake, taking a shortcut to the beneficial predator deterrence.
But wonderful things can emerge as well: mimicry can be a part of mutualistic relationships that benefit both the model and the mimic, demonstrating productive sharing of a “language” across evolutionary lineages.
The general idea
Müllerian mimicry is a well-studied phenomenon, particularly in butterflies, in which completely unrelated species come to display the same patterns and coloration. Each species is unpalatable to eat for different reasons, but all have come to share the same visual cues to make this point known to their common predators. As a group, sending the most consistent signal to predators achieves the best result – not being eaten – for all members.
Mimics of the gut
The ways in which humans host and depend on the collection of bacteria in our gut, our microbiome, is another intimate example of mutualism. Improbably, a new study has shown that gut bacteria have learned to speak the complex language of our immune system, mimicking and modifying a pro-inflammatory signal into a signal for immune suppression, directly protecting us from the damage caused by autoimmune disorders in which the body attacks itself.
The human immune system is a network of cells distributed throughout each of our bodies. Populations of immune cells travel through our bloodstreams and through our lymphatic systems, constantly interacting and exchanging information which results, in the best cases, in keeping us healthy. In terms of our immune systems, healthy implies a delicate balance between immune activation and suppression. Our immune system needs to be sensitive to real threats, but in their absence, it needs to be reined in to prevent damage to our own cells and tissue.
Think of the immune system as two major groups of cells: surveillance cells and decider cells. Surveillance cells, called Antigen Presenting Cells, or APCs, rove around the body and physically engulf any cellular debris that they find – including whole bacterial cells. They digest it into tiny fragments, and then decorate their exterior surface with fragments of what they’ve found. Then they present their findings to their more discerning decider cells. The important decider cells in this case are called T cells, and the decision to mount an immune attack based on the information they receive lies with them.
The handoff between APCs and T cells is a crucial point of communication where details matter. T cells are trained to distinguish fragments of foreign proteins – like ones from harmful bacteria – from fragments of human proteins. They do this using the receptors on their surface, which are specific for different protein fragments the way locks require specific keys. When the key fits, our immune system becomes activated until the threat can be resolved.
T Cell
Fragments of your body's own proteins are part of the cellular debris vacuumed up by APCs. Fortunately, two systems are in place to prevent those fragments from triggering immune activation. First, the T cells with matching receptors are very rare in the total T cell population. Second, in a few cases, protein fragments do match, but interact with the T cell's receptor in a weaker way. These weak binding interactions signal to the T cell to suppress immune activity.
Local immune suppression sounds risky, but it's the perfect antidote when the immune system accidentally gets activated by a human protein. The cellular damage caused by immune activation often releases more human proteins into the milieu, triggering more activation, in a positive-feedback cycle. Inflammatory Bowel Disorders (IBD) are thought to arise from a variation on this, where immune cells are overly prone to attack our friendly, commensal gut bacteria.
Speaking in proteins
A remarkable study by co-first authors Roopa Hebbandi Nanjundappa and Francesca Ronchi from the University of Calgary describes a common human gut bacteria that floods the immune system signaling center with a mimic of a host protein-fragment with that rare, immune suppressing ability. By following all the existing rules of the language of protein fragment and receptor interaction, a gut bacterium sends a signal that ultimately protects us from autoimmune damage.

It's engimatic
In the United States, Bacteroides species are the most abundant group of bacteria in the human gut. By searching the genomes of Bacteroides species, the researchers found that many Bacteroides expressed a protein that had a tiny piece in common with a human protein. In a mouse model, mice suffered significantly more damage from IBDs when their Bacteroides lacked the fragment, suggesting that Bacteroides species provide a natural immune suppressant that protects both us and our commensal gut bacteria from autoimmune attack.
Bacteria, due to their rapid growth rate, can evolve and potentially change their genetic material much more rapidly than humans, but mutation rate alone cannot explain the origin of such a spectacularly lucky protein fragment in Bacteroides. In fact, all bacterial genomes harbor even smaller genetic elements which often have their own mechanisms to rapidly spread through populations, significantly contributing to bacterial genetic diversity. The Bacteroides protein that is the source of the human fragment mimic is encoded within one such genetic element, called a transposon, which has jumped into the genomes of multiple Bacteroides species.
Although the transposon protein had (or still has) an original function completely distinct from the human protein that it mimics, as part of the Bacteroides genome where it now lives it contributes not only to its direct host’s physiology but to human physiology, through its fortuitous engagement with the language of our immune system.
The dense network of interactions that exists unseen between us and our microbiome is rapidly being charted. An advantage to hosting trillions of bacteria is that they rapidly explore genetic and structural space: at the end of the day, the functions that benefit us and our microbiome, by limiting disease and restoring balance to our immune system, are the ones most likely to last. The bacteria in our microbiome have spent centuries identifying and learning to pull the levers of our immune system. For those interested in designing effective therapies for autoimmune disorders, there is plenty to learn from them.


Which came first, the microbe or the immunity? The microbe, of course. Considering the evolutionary timescale, mutualism developed with a large degree of synchronicity. Microbes have spent not just centuries but millions (billions?) of years cohabiting with animal and plant hosts, so it's hard to say what any given gene originally did.
In the paper's introduction the authors state, "this T cell specificity might have been evolutionarily maintained because it was beneficial to the host." I wonder how far back that particular T cell receptor's genetic conservation could be traced phylogenetically.
Having said all that, this is a really neat piece of research to highlight because it shows a mechanism through which that tolerance is achieved, in which the associated T cells calm down and block the pro-inflammatory cascade. This hints that there could be many other lower-abundance proteins that could mimic mammalian epitopes – the sequence of amino acids in the protein fragment that the APCs display to the T cells – combining to enforce a certain level of inflammation.
Could we reverse-engineer our own desired levels of immune activation? The article asserts that these T cells could be therapeutic in IBD, but what about the bacteria that have the transposon? Lots of cool possibilities; hopefully the ever-accelerating field of proteomics can reveal more immune-regulatory epitopes shared between mammals and their microbiome.