How seven cases of a mysterious opioid-induced disease revolutionized Parkinson's research
In the early 1980s, seven people took synthetic heroin. What happened next drastically changed our understanding of Parkinson's disease, and how to treat it
In the early 1980s, doctors across northern California encountered patients with curious symptoms. From Watsonville to San Jose, young people were experiencing a combination of visual hallucinations, jerking limbs, and stiffness. Their abilities to move and speak worsened over a space of mere weeks, and by the time they sought medical help they had become totally immobile.
In San Jose's Santa Clara Valley Medical Center, psychiatrists insisted that their medical mystery patient had a neurological disorder. However, neurologists were adamant that the patient was undergoing a psychiatric episode. Amidst the arguments, Director of Neurology, William Langston, noticed a striking resemblance of the patient's condition to that of Parkinson's disease (PD). They were alert, but their body was rigid – with an arm involuntarily raised for long periods of time. PD is the second most common neurodegenerative disorder (behind Alzheimer's disease), and the most common neurodegenerative movement disorder worldwide, but it is typically associated with old age, and symptoms develop over a period of years – not weeks.
The doctors used news media alerts and police assistance to see if their medical mystery patient was an isolated incident. Soon, they discovered six other similar cases across the state. The patients did not know each other, but all had one thing in common: they had recently taken the same recreational drug. It was described as a "synthetic heroin" and, after police raids and cooperation from local dealers, chemical analyses of the drug revealed that one batch was near pure 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). MPTP is a byproduct of the synthesis of an opioid, with effects similar to morphine. Our bodies metabolize MPTP into a toxin that selectively kills neurons in the substantia nigra, the same area of the brain affected in PD.
A nearly identical case of synthetic opioid-induced Parkinsonism had actually been identified a few years previously but had gone unnoticed by much of the scientific community. It wasn't until Langston's team published their findings, reporting that MPTP consumption led to their patients' Parkinsonism before PD researchers took notice.
Within hours of the publication of Langston's paper, all of Aldrich Chemical's (one of the largest retailers of chemical supplies) commercial stock of MPTP had sold out. The cost of MPTP increased almost 100-fold once it was restocked. The doctors had not realized it at the time, but they had just informed the world of a valuable new research model of PD.
Human diseases like PD are often difficult to model in a laboratory setting. Before new drugs are eligible for human trials, they must first be tested on cells or animals. However, in order to evaluate the effectiveness of a drug, a disease-like state must be induced in these cells or animals first. Therefore, the striking clinical and pathological similarities of the effects of MPTP consumption to PD, though tragic for the patients, was a silver lining for research. Just a year after publishing their findings, other scientists had reported the remarkable effects of MPTP in non-human primates. Like in humans, MPTP induced motor symptoms in monkeys and these symptoms were alleviated with levodopa – the precursor to dopamine and a common PD treatment. Levodopa-responsiveness is usually used in the clinical diagnosis of PD.
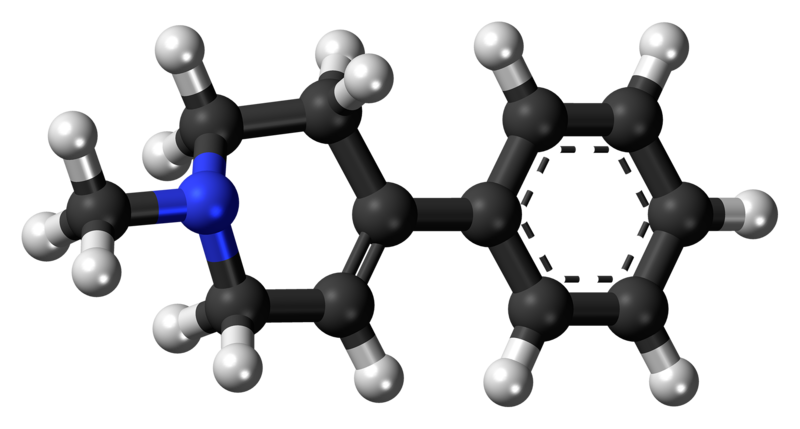
The chemical structure of MPTP
Jynto on Wikimedia Commons
In the near-forty years since its discovery, over six thousand MPTP-related PD studies have been published. The majority of these have been performed in mice – interestingly, rats, a common neuroscience lab model, are resistant to the effects of MPTP. The MPTP mouse model has been useful in picking apart the mechanisms of disease, especially the effects of mitochondrial dysfunction in PD.
These animal models have also informed the development of potential therapeutics. Substances that could prevent MPTP-induced neurodegeneration and movement symptoms in animals could also be protective in human PD. For example, one study found inhibitors of an enzyme called monoamine oxidase (MAO) B, were protective against the toxic effects of MPTP in mice. This discovery inspired the first clinical trials for MAO B inhibitors for treating PD, known as selegiline and rasagiline. Both of these drugs are prescribed to PD patients today, however, like all PD treatments, they unfortunately only improve symptoms rather than modify the severity or progression of the disease.
In addition to helping scientists develop PD drugs, MPTP animal models have also been useful in understanding the environmental risk factors of PD. The toxic metabolite of MPTP, MPP+, has a very similar structure to a herbicide called paraquat. Paraquat is used worldwide, under the brand name Gramoxone. However, many studies have now demonstrated an association between paraquat use and PD development. While some countries, including the EU nations, have now banned its use due to this link with PD, paraquat remains one of the most widely used herbicides today.
The unfortunate case of the "Frozen Addicts" has been an enormous breakthrough for the PD field in countless ways. Their misfortune has led to the development of multiple therapies and increased our understanding of the disease, undoubtedly aiding the lives of millions of PD patients across the world and will continue to do so.


I really enjoyed this article. It’s a great insight into how important reporting and publicizing unusual findings are to medical research. This story actually reminded me of the New Yorker article 1 which, as part of a larger article on anti-viral therapeutics, chronicled the history of linking certain rare and unusual cancers to retroviruses at the start of the AIDS crisis. The idea that viruses could cause cancers was revolutionary (and not immediately accepted!) but it has lead to diagnoses and treatments which have saved countless lives.