Heather Hazzan/SELF Magazine via Flickr
Why do the COVID-19 mRNA vaccines need to be kept so cold?
The Pfizer and Moderna vaccine cold chains, explained
Monday, December 14th, marked the first public vaccinations of the Pfizer-BioNTech vaccine in the US. It came 339 days after the release of the SARS-CoV-2 genome and months of clinical trials involving more than 43,000 people during a raging pandemic that has infected 73,600,000 and killed 1,640,000 people worldwide. Pfizer-BioNTech’s vaccine is among 23 vaccines in the final stages of testing and approval – phase 3 and/or nearing approval. In the United States, Moderna’s vaccine is expected to be distributed soon, after endorsement by an FDA panel, and emergency approval received, with similar efficacy results to Pfizer-BioNTech’s vaccine.
Both of these vaccines are RNA-based. Instead of priming the immune system with a dead virus or a piece of a virus, as vaccines in the past have done, these vaccines deliver a template – RNA – for our cells to make a single protein from SARS-CoV-2. The body creates the protein, generates a protective immune response, then throws away the RNA – much like kids passing notes in class, then crumpling them up and tossing them in the trash.
Each vaccine consists of two doses, given a few weeks apart. Manufacturing is expected to produce close to 2 billion doses in 2021 (up to 1.3 billion for Pfizer-BioNTech, up to 1 billion for Moderna), enough for almost 15% of the world’s population if these companies reach their maximum output. Despite unprecedented success in the development of these new vaccines, their extensive testing, and scaling up manufacturing, there remains another hill to climb before these vaccines can help protect people. These RNA vaccines require extremely cold temperatures.

A common ultracold freezer found in a lab. These types of freezers can cost $10,000 or more
Pleple2000 via Wikimedia
Pfizer-BioNTech’s vaccine requires -70°C (-94°F), colder than the South Pole, and only lasts around 5 days once placed in a refrigerator. Moderna’s vaccine is a bit more forgiving, shipped at -20°C (-4°F) and good for a month in a refrigerator. For comparison, inactivated or live attenuated vaccines (like the flu vaccine) are stored at typical refrigerator temperatures, about 2-8°C (46°F). While the Sputnik V vaccine also needs freezer-like temperatures, we can expect emerging vaccines from Novavax (NVX-CoV2373), AstraZeneca-University of Oxford (AZD1222), Johnson & Johnson (Ad26.COV2.S), and others to require only refrigerator temperatures.
RNA is notorious for its instability. Before working with RNA, it’s common for scientists working with it in a lab to clean their workspace of “RNases” or ubiquitous environmental enzymes that break down RNA quickly. Unlike DNA, which typically consists of two strands and contains a submolecule called deoxyribose (hence “DNA”), RNA is single-stranded and contains ribose (hence “RNA”), which makes RNA molecules more susceptible to degradation. Despite the chemical modifications and packaging these companies use to make it more resistant, it still suffers compared to DNA or protein.
The extreme temperatures slow down chemical and enzymatic degradation, allowing the RNA vaccines to maintain their efficacy from manufacturing to injection. RNA’s instability is good for our cells: many copies of RNA are transcribed from DNA templates, which are then used to create proteins. But as the proteins required by the cell are made, the instructions are degraded away.
These companies define these temperatures through testing the vaccine across a range of temperatures and storage durations. Protein vaccines have their own requirements as well. While they are typically stored in a refrigerator, freezing them can disfigure the proteins, rendering them ineffective. Pfizer-BioNTech has mentioned ongoing studies on storage conditions, but for now, ultracold transportation with GPS tracking it is.
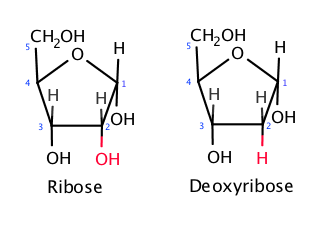
The only chemical difference between RNA and DNA is the presence of a single oxygen atom (RNA carries a ribose, left, highlighted in red) that is missing in DNA, which carries a deoxyribose (right)
Miranda19983$! via Wikimedia
The transport of vaccines, and cold items more broadly, from a manufacturing site to a recipient is known as the cold chain. The cold chain can be divided into different temperature requirements, such as freezer temperatures (about -20°C) that the Moderna vaccine requires or ultracold temperatures for Pfizer-BioNTech’s, or even cryogenic temperatures which can reach -150°C (-238°F) for shipping biological materials. Over the course of the first week, 636 sites were expected to receive the vaccine through the Fedex and UPS shipping networks. Pfizer’s shipping containers can stay cold enough for 10 days unopened. After receiving these precious vaccines, sites need to maintain these temperatures in ultra-low freezers (good for six months), which cost well over $10,000 with enormous energy consumption, in the original shipping container by refilling with dry ice (-79°C, -109°F, good for 30 days) or storing in a refrigerator, where the vaccines must be used within 5 days.
This extreme cold chain has sprouted intense investment and concern. Perhaps most notably, India has projected needing millions of additional equipment to handle these various vaccines. The dramatic need for cold chain solutions across the globe has grabbed Wall Street’s attention, driving investment in one company, Cryoport, up over 200% year to date. Given the challenges in countries with developed healthcare infrastructures, it’s not hard to imagine the additional challenges in less developed countries. There’s some precedent before with the Ebola vaccine, but the scale of these infections are vastly different. Despite these challenges, perhaps the first issue is the availability of these vaccines, which was quickly cleared by Western countries.
The cold chain required for vaccines is not a new challenge. The WHO and PATH, among others, have been focusing on supply chain issues for decades, developing new technology like solar-powered refrigerators and calling for action in 2014. The overwhelming scale of COVID-19 and mass vaccination campaigns mixed with subfreezer temperatures is a new challenge. As we end 2020, we can hope more efficacious vaccines are approved early in 2021, allowing more people from more walks of life to have access to protection against COVID-19, ending the pandemic.
